O2875
Orotic acid potassium salt
≥98%
Synonym(s):
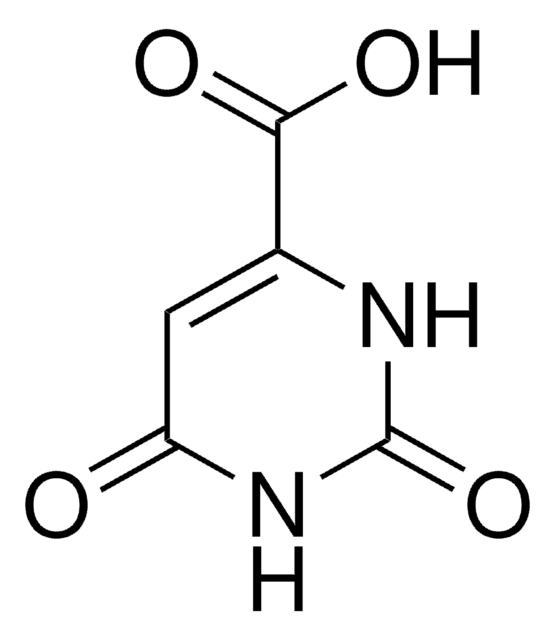
6-Carboxy-2,4-dihydroxypyrimidine, Uracil-6-carboxylic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
C5H3N2O4K
CAS Number:
Molecular Weight:
194.19
EC Number:
MDL number:
UNSPSC Code:
41106305
PubChem Substance ID:
NACRES:
NA.51
Recommended Products
Quality Level
Assay
≥98%
form
powder
solubility
1 M NaOH: 25 mg/mL, clear to slightly hazy, colorless to faintly yellow
SMILES string
[K+].[O-]C(=O)C1=CC(=O)NC(=O)N1
InChI
1S/C5H4N2O4.K/c8-3-1-2(4(9)10)6-5(11)7-3;/h1H,(H,9,10)(H2,6,7,8,11);/q;+1/p-1
InChI key
DHBUISJCVRMTAZ-UHFFFAOYSA-M
Application
Orotic acid (OA) is an intermediate in de novo pyrimidine biosynthesis that may be used to study the specificity and kinetics of orotate phosphoribosyltransferase (OPRT) which catalyzes the reversible phosphoribosyl transfer from 5′-phospho-α-d-ribose 1′-diphosphate (PRPP) to orotic acid (OA), forming pyrophosphate and orotidine 5′-monophosphate (OMP). Orotic acid is used as a starting material for the potential commercial bioproduction of uridine 5′-monophosphate (UMP) by microbes such as Corynebacterium ammoniagenes (ATCC 6872) or Saccharomyces cerevisiae. OA may be used to study the AMPK/SREBP-1 dependent cell signaling pathway and transcription regulation mechanisms that induce hepatic lipogenesis.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service