D4641
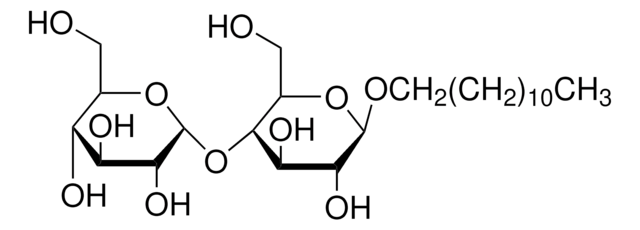
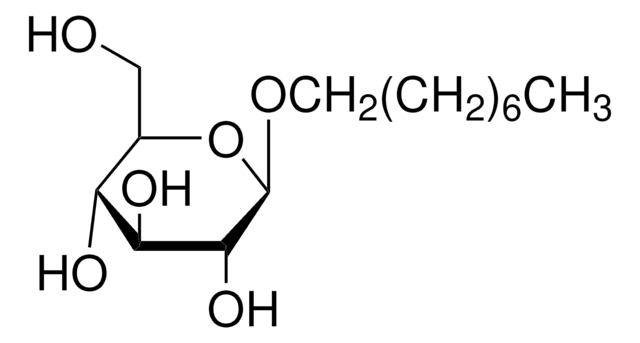
n-Dodecyl β-D-maltoside
≥98% (GC)
Synonym(s):
DDM, Lauryl-β-D-maltoside
About This Item
Recommended Products
biological source
corn
Quality Level
description
non-ionic
assay
≥98% (GC)
form
powder
mol wt
micellar avg mol wt 50,000
aggregation number
98
technique(s)
protein quantification: suitable
impurities
<1.5% water (Karl Fischer)
CMC
0.15 mM (20-25°C)
mp
224-226 °C (lit.)
solubility
water: 50 mg/mL, clear to very slightly hazy, colorless
storage temp.
−20°C
SMILES string
CCCCCCCCCCCCO[C@@H]1O[C@H](CO)[C@@H](O[C@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)[C@H](O)[C@H]1O
InChI
1S/C24H46O11/c1-2-3-4-5-6-7-8-9-10-11-12-32-23-21(31)19(29)22(16(14-26)34-23)35-24-20(30)18(28)17(27)15(13-25)33-24/h15-31H,2-14H2,1H3/t15-,16-,17-,18+,19-,20-,21-,22-,23-,24-/m1/s1
InChI key
NLEBIOOXCVAHBD-QKMCSOCLSA-N
Looking for similar products? Visit Product Comparison Guide
General description
In addition to its primary function, N-dodecyl-β-D-maltoside has diverse applications, including the purification and stabilization of RNA polymerase, the detection of protein-lipid interactions, and serving as a substrate for glucosyl and xylosyl transfer by glycogenin. Its mild and non-denaturing properties have further led to its utilization in protein-anesthetic studies. Overall, N-dodecyl-β-D-maltoside emerges as a versatile detergent with a broad range of applications in membrane protein research and beyond. Its pivotal role in preserving protein structure and function establishes it as an indispensable tool for exploring the intricacies of membrane biology.
Application
Features and Benefits
- Highly versatile surfactant for your cell biology and biochemical research
- Suitable for protein quantification
Other Notes
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service